Microscope analysis for soil health
Soil health is a complex subject and there are many ways to approach it. Biodiversity is a key aspect of soil health with profound impacts on agricultural success and sustainability. Each organism has specific functions that affect the rest of the soil ecosystem, including plants.
Some key functions of a healthy soil ecosystem include:
Good water retention and drainage
Healthy structure and resistance to erosion
Richer, more diverse nutrient cycling and retention
Improved plant health
Increased carbon storage
Resilience against pest and disease outbreaks
The goal of this analysis is to develop a profile of the soil’s ecological status, which considers diversity, the physical characteristics of the soil habitat, and where possible takes into account outside factors such as agricultural activities that can affect, and be affected by, the soil ecosystem.
Observing soil in the microscope can provide great insight into the current ecological status, changes over time or with treatments, and the effectiveness of soil management strategies.
The method
To observe soil in the microscope, samples are mixed with water and then allowed to rest for two days. They are then viewed at 100x and 400x magnification. Bacteria, protozoa, and fungi are observed and recorded along with physical characteristics of the soil and any other organisms that may be present, such as nematodes. Healthy soil should have many different kinds of organisms with populations that are in balance with one another. There should also be visible evidence that the physical habitat supports a complex ecosystem.
Groups of organisms observed with the microscope
The main groups of organisms considered during a microscope analysis are bacteria, fungi, and protozoa. Other organisms that are sometimes observed include nematodes and rotifers.
Bacteria

Bacteria are very small (1 μm), but it is possible to see them at 400x magnification. Bacteria cannot be specifically identified using only a microscope, but we can estimate the abundance and basic characteristics such as spiral, rod, or round shapes, and the type of movement they have, which all give clues about bacterial diversity.
For this analysis, any noticeable signs of bacterial activity and diversity are written down, and an estimate of bacterial biomass is made. This estimate is then compared against the fungal biomass estimate (next section) to determine whether the sample is dominated by bacteria or fungi, or equally balanced. For most agriculture and garden soils, the recommendation is to aim for a 1:1 balance of bacteria and fungi.
Fungi
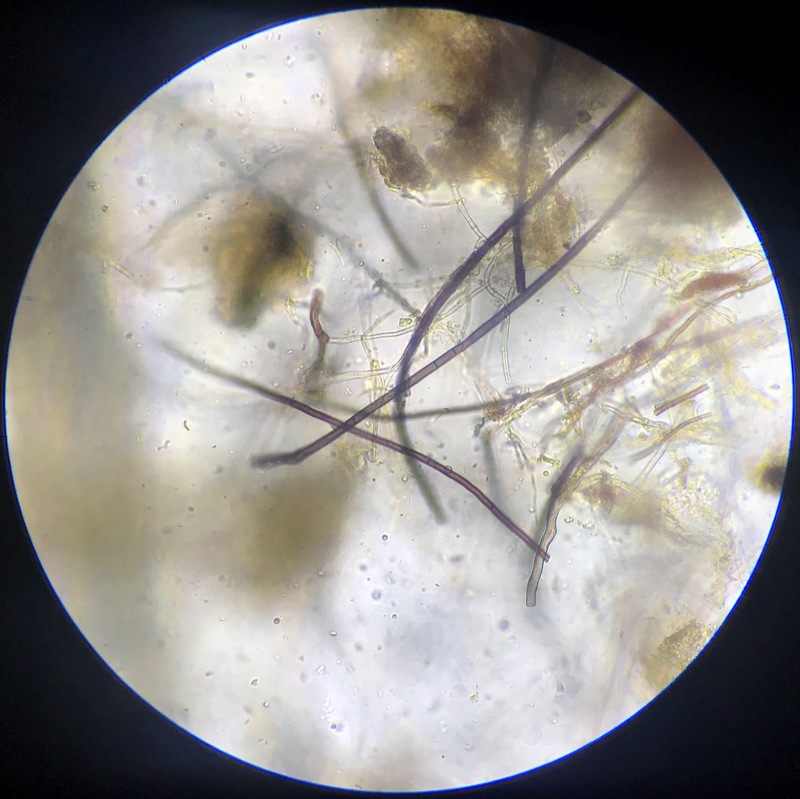
Healthy soil typically has robust networks of diverse fungal threads called “hyphae”. In the microscope, these look like clear or brown strands, typically between 2-6 μm in diameter. The length of fungal hyphae varies greatly in prepared samples, and long, robust strands are considered a sign of good conditions in the soil. When disturbance is minimal, fungal networks weave through the soil, extracting nutrients and interacting with plants. They help bind particles together into aggregates, and they provide significant benefits to plants through the exchange of nutrients and much more. Soil fungi develop slowly and are particularly sensitive to disturbance and other stressors, which makes them excellent indicators of the soil’s ecological status. In the microscope, the presence of septate fungal hyphae wider than 3 μm is considered a sign of good growing conditions. Large numbers of light, thin hyphae could be a sign of dense, oxygen-poor soil, which is a less supportive ecosystem for most agricultural crops.
In analysis reports, the fungal biomass estimate is compared with the number of individual fungal hyphae fragments to provide a simple numerical score (total evaluation of fungi / totalvurdering sopp) on a scale of 0-5, where 0 is very poor and 5 is very high. This simplified score is particularly useful for tracking changes over time, or to compare the effect of treatments or soil management techniques.
Protozoa
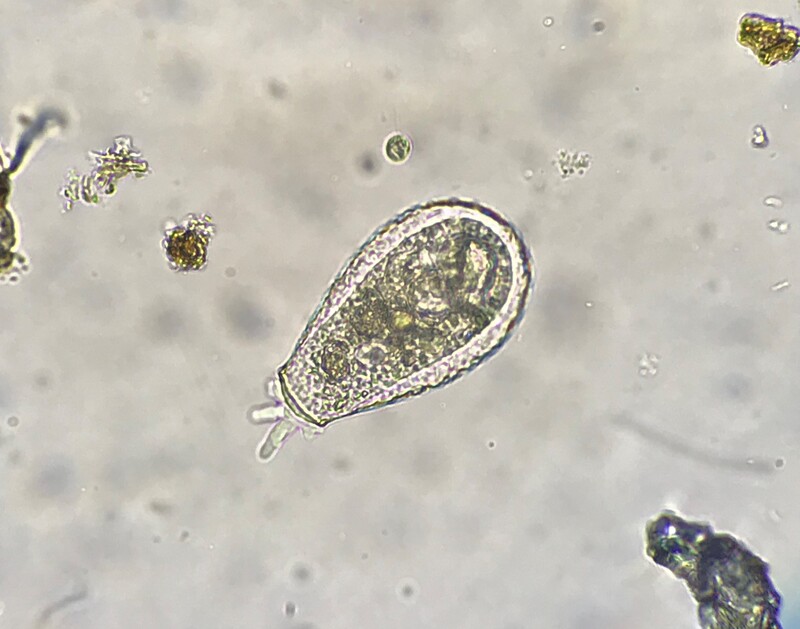
Protozoa are an incredibly diverse group of single-celled, eukaryotic organisms, which have a predatory role in the soil food web. The role of protozoa in agriculture tends to be underappreciated, perhaps because they are difficult to study, but they are critically important members of the soil food web. For example, bacteria tend to consume a lot of nitrogen and store it in their bodies, but protozoa have little need for nitrogen, so when they consume bacteria they release what they don’t need back into the soil in a form that plants can easily use. Protozoa are voracious predators of bacteria, but they are selective about which species they consume. Each species of protozoa feeds on particular types of bacteria, and in doing so they each play specialized roles in the soil community. This could also mean that protozoa diversity may be an indicator of bacterial diversity in the soil. Protozoa have also been found to promote plant health and disease resistance and improve growth independently of nutrients.
When evaluating protozoa in soil samples, diversity and balance are the top priorities. The ideal sample will have moderate representation from as many groups as possible, with good diversity within those groups. Since it is not feasible to identify all the species of protozoa in a routine analysis, they are instead grouped according to easily observable characteristics. This provides an efficient way to estimate diversity in living, active samples.
We have attempted to create an index that takes into account both the number of groups and the number of organisms within each group. This index is a work in progress, but generally speaking, a more positive result will have several groups and relatively even distribution of individuals within the groups.
What can we do to support soil life?
It is important that we remember to view soil as a habitat and an ecosystem, and to shift our mindset from feeding plants to feeding the soil, which will in turn feed the plants and support them in many other ways.
Here is a summary of the basic conditions that favour beneficial microbial activity in soil and how to provide them:
Microbes need: |
You can provide it by: |
|---|---|
Moisture |
Keeping the soil covered |
Oxygen |
Allowing natural structure to develop; avoid compaction |
Energy and nutrients |
Maintaining good cover with living plants and mulch; as much diversity as possible |
Shelter |
Keeping the soil covered |
Reduced disturbance |
Minimizing tillage, driving, and chemical interference |
Earthworm activity* |
All of the above |
*Earthworms are known as “ecosystem engineers”. Their activity improves soil quality and creates conditions that support beneficial microorganisms.
Leaving soil undisturbed is one of the most important ways we can support soil life. Take a closer look at the effect of leaving garden soil undisturbed, vs. preparing it using a conventional soil preparation method in this article: Comparing dig vs. no dig soil in a market garden.
Suggestions for further reading:
Bender, S. Franz et al. "An Underground Revolution: Biodiversity And Soil Ecological Engineering For Agricultural Sustainability". Trends In Ecology & Evolution, vol 31, no. 6, 2016, pp. 440-452. Elsevier BV, doi:10.1016/j.tree.2016.02.016.
Bonkowski, M., and F. Brandt. "Do Soil Protozoa Enhance Plant Growth By Hormonal Effects?". Soil Biology And Biochemistry, vol 34, no. 11, 2002, pp. 1709-1715. Elsevier BV, doi:10.1016/s0038-0717(02)00157-8.
Bonkowski, M. (2004). Protozoa and plant growth: the microbial loop in soil revisited. New Phytologist, 162(3), pp.617-631.
Brussaard, Lijbert. “Biodiversity and Ecosystem Functioning in Soil.” Ambio, vol. 26, no. 8, 1997, pp. 563–570. JSTOR, www.jstor.org/stable/4314670.
Cardon, Z. and Whitbeck, J. (2019). The Rhizosphere. Academic Press.
Cobb, N. (1914). Yearbook of the Department of Agriculture. pp.457-490.
Davies, N., Stoker, D., Windsor, D., Ashcroft, M., Coburn, M. and Andrews, W. (1973). A Guide to the Study of Soil Ecology. Scarborough: Prentice-Hall of Canada, Ltd.
Lowenfels, J. (2013). Teaming with Nutrients. Timber Press.
Malik, A., Chowdhury, S., Schlager, V., Oliver, A., Puissant, J., Vazquez, P., Jehmlich, N., von Bergen, M., Griffiths, R. and Gleixner, G. (2016). Soil Fungal:Bacterial Ratios Are Linked to Altered Carbon Cycling. Frontiers in Microbiology, 7.
PAUL, E. (2017). SOIL MICROBIOLOGY, ECOLOGY AND BIOCHEMISTRY. 4th ed. [S.l.]: ELSEVIER ACADEMIC PRESS.
Rosenberg, K., Bertaux, J., Krome, K., Hartmann, A., Scheu, S. and Bonkowski, M. (2009). Soil amoebae rapidly change bacterial community composition in the rhizosphere of Arabidopsis thaliana. The ISME Journal, 3(6), pp.675-684.
Shin, W., Islam, R., Benson, A., Joe, M., Kim, K., Gopal, S., Samaddar, S., Banerjee, S. and Sa, T. (2016). Role of Diazotrophic Bacteria in Biological Nitrogen Fixation and Plant Growth Improvement. Korean Journal of Soil Science and Fertilizer, 49(1), pp.17-29.
Sturz, A. and Christie, B. (2003). Beneficial microbial allelopathies in the root zone: the management of soil quality and plant disease with rhizobacteria. Soil and Tillage Research, 72(2), pp.107-123.
Weidner, S., Latz, E., Agaras, B., Valverde, C. and Jousset, A. (2016). Protozoa stimulate the plant beneficial activity of rhizospheric pseudomonads. Plant and Soil, 410(1-2), pp.509-515.
Winding, A., Rønn, R. and Hendriksen, N. (1997). Bacteria and protozoa in soil microhabitats as affected by earthworms. Biology and Fertility of Soils, 24(2), pp.133-140.